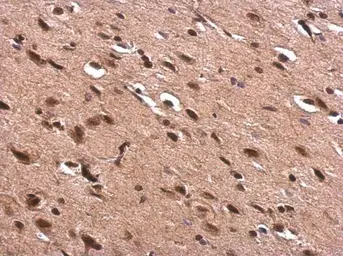
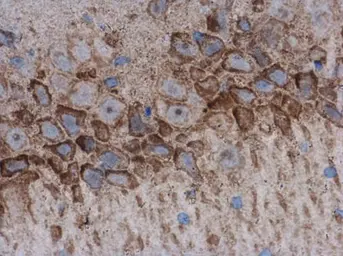
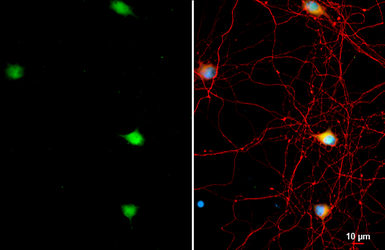
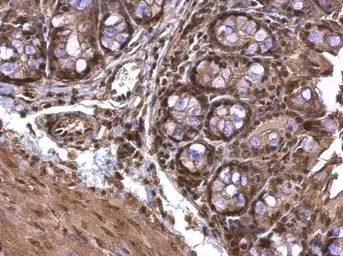
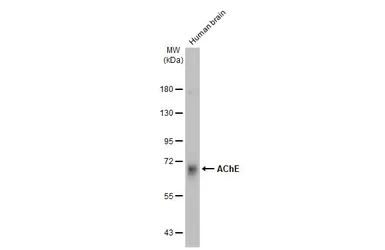
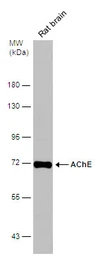
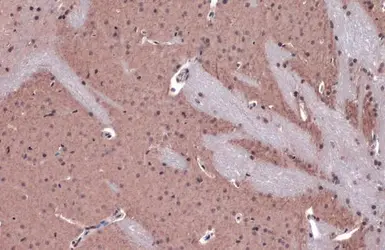
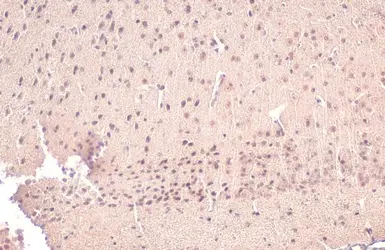
AChE antibody
Cat. No. GTX101648
Cat. No. GTX101648
-
HostRabbit
-
ClonalityPolyclonal
-
IsotypeIgG
-
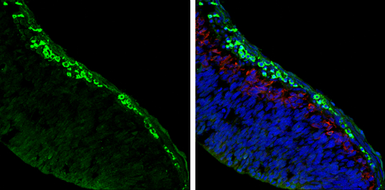
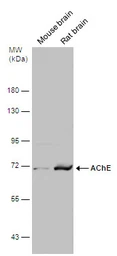
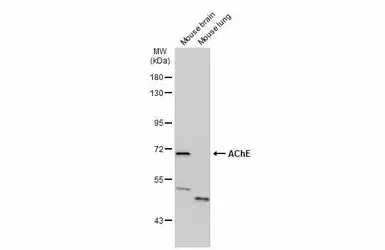
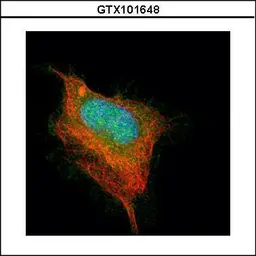
ApplicationsWB ICC/IF IHC-P IHC-Fr
-
ReactivityHuman, Mouse, Rat