About Us




GeneTex creates reagents for biomedical research, assay development, and in vitro diagnostics. Its products are made to standards outlined in quality management systems ISO 9001:2015, ISO 13485:2016, and ISO 14001:2015 are supported by comprehensive customer service.
GeneTex was founded in San Antonio, Texas in 1997 in an effort to more efficiently manage requests for a number of highly regarded and well-published antibodies generated in the labs of the company’s creators. In 2007, to increase its manufacturing capabilities, GeneTex Taiwan was established. Two years later, GeneTex’s U.S. office relocated to Irvine, California to be close to the many major universities, biotech companies, and research institutes in that region of the country. In 2020, GeneTex introduced new facilities for recombinant monoclonal antibody and recombinant protein production in combination with new technologies to improve manufacturing efficiency and quality control. Currently, GeneTex has more than 150 employees, of which 70% have postgraduate degrees.
-

GeneTex founded in San Antonio, TX
-

New production and development facilities opened in Taiwan
-

GeneTex U.S. relocates to Irvine, CA
-

Recombinant antibody and protein production facilities opened in Taiwan
-

Over 150 employees in three locations
Our Antibody Technology
-
Learn morePolyclonalAntibodies
-
Learn moreMonoclonalAntibodies
-
Learn moreRecombinantsAntibodies
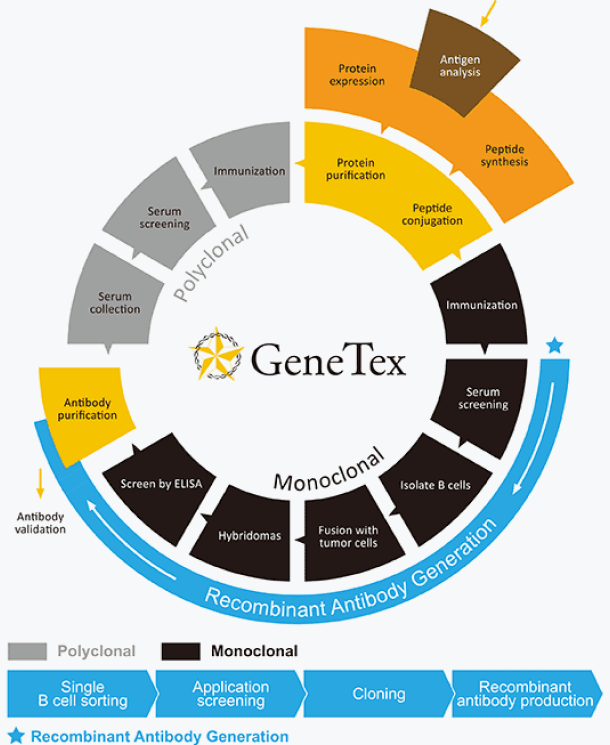
While commercial polyclonal and monoclonal antibodies have driven progress in science and medicine since their introduction, the generation of reliable antibodies remains an essential goal in the quest for data accuracy and reproducibility. On the forefront of high-quality antibody manufacturing, GeneTex has successfully incorporated recombinant monoclonal antibody technology into its product development platform. This technology has numerous well-documented advantages, with consistency of performance being of paramount importance. GeneTex’s capability in this area enhances its position in the research community and the biotechnology industry and spearheads its efforts to provide the most trusted antibody products available.
Our commitment to provide our customers with reliable reagents and to drive life science research reflects the philosophy and approach we employ when we create our products.
Using meticulous manufacturing procedures and stringent testing, we have produced and validated a comprehensive collection of antibodies and research tools that researchers use each day to expand biomedical knowledge.
After more than 25 years of producing quality reagents, GeneTex continues to distinguish itself by emphasizing reagent validation. Poorly characterized antibodies remain an industry-wide challenge that impacts progress in the life sciences and biomedicine.
At GeneTex, product reliability is our primary concern. Stringent protocols have been established to validate our antibodies for various applications, including WB, IP, IHC, ICC/IF, FACS, and ELISA, using cell lines with known target protein expression levels and a diverse array of tissue samples. Lot-to-lot antibody performance is maintained through a combination of fully characterized reference lots, clone verification, and thorough quality control.
Founded by scientists for scientists, we at GeneTex understand that researchers expect and demand product fidelity. With that in mind, we are increasingly incorporating more Knockout/Knockdown (KO/KD) validation testing into our quality assurance (QA) regimen. KO/KD testing is a major pillar of our enhanced “Five-Pillar Validation” program with the goal of demonstrating to our customers that the GeneTex reagents they purchase are worthy of their trust.


There has been growing distrust of commercially available antibodies with regard to specificity and performance variability. As a result, the research community has demanded that antibody suppliers increase their efforts to more thoroughly validate their products. One approach to address this is through knockout or knockdown of the target protein.
At GeneTex, we continue to provide the research community with the highest quality and most stringently validated antibodies by utilizing CRISPR/Cas9- and shRNA-generated cell lysates for our western blot assays. To date, we have evaluated most of our featured products and will continue to expand these efforts throughout our entire antibody catalog.
Our Emissions Reduction Efforts
GeneTex's emissions reduction target is to achieve a 42% decrease in combined Scope 1 and Scope 2 emissions by 2034 from our 2024 baseline.