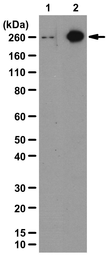
Acetyl-CoA Carboxylase 1 (phospho Ser79) antibody
Cat. No. GTX30764
Cat. No. GTX30764
-
HostRabbit
-
ClonalityPolyclonal
-
IsotypeIgG
-
ApplicationsWB
-
ReactivityHuman