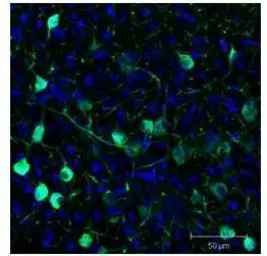
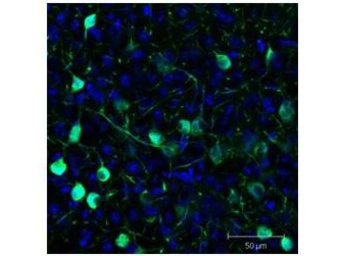
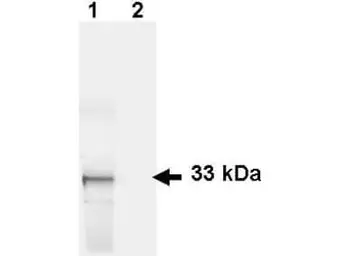
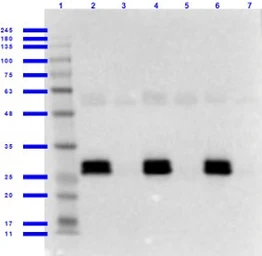
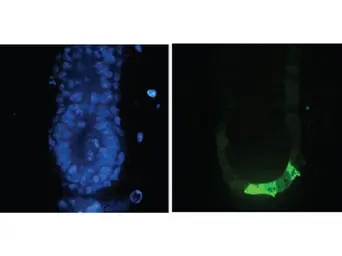
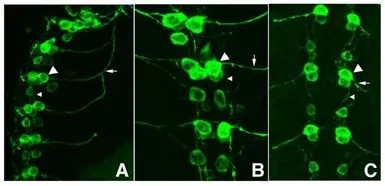
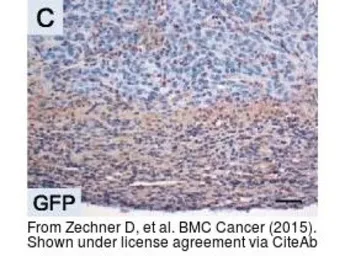
GFP antibody
Cat. No. GTX26673
Cat. No. GTX26673
-
HostGoat
-
ClonalityPolyclonal
-
IsotypeIgG
-
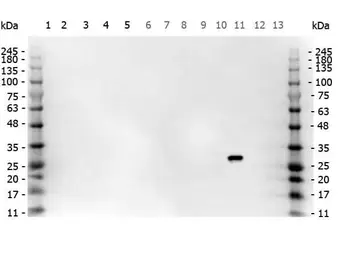
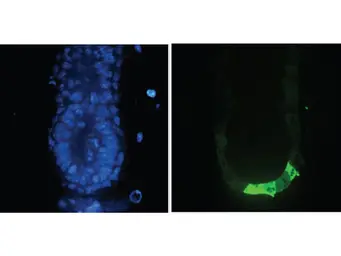
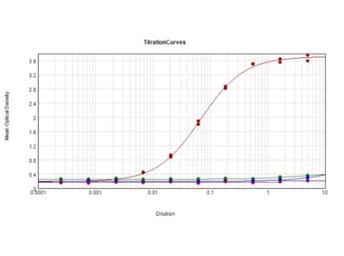
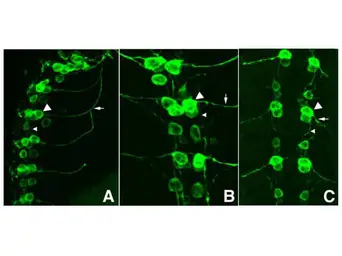
ApplicationsWB ICC/IF IHC-P IHC-Fr FCM IP ELISA IHC Multiplexing Purification IHC (Free Floating)
-
ReactivitySpecies independent