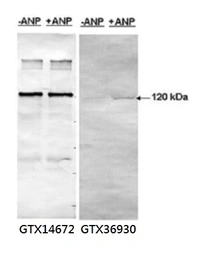
PDE5A (phospho Ser 92) antibody
Cat. No. GTX36930
Cat. No. GTX36930
-
HostRabbit
-
ClonalityPolyclonal
-
IsotypeIgG
-
ApplicationsWB ICC/IF IP ELISA IHC
-
ReactivityHuman, Mouse, Rat, Zebrafish, Sheep, Bovine, Pig