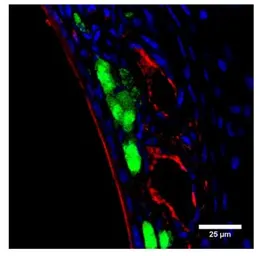
PGCB antibody
Cat. No. GTX14784
Cat. No. GTX14784
-
HostRabbit
-
ClonalityPolyclonal
-
IsotypeIgG
-
ApplicationsWB ICC/IF IP ELISA IHC
-
ReactivityHuman, Mouse, Rat