Rat PAM protein, His tag (active)
Cat. No. GTX00374-pro
Cat. No. GTX00374-pro
-
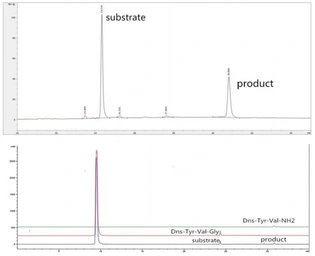
ApplicationsFunctional Assay
-
SpeciesRat