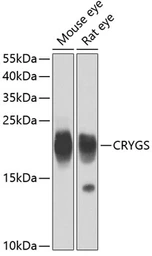
Beta crystallin S antibody
Cat. No. GTX32540
Cat. No. GTX32540
-
HostRabbit
-
ClonalityPolyclonal
-
IsotypeIgG
-
ApplicationsWB
-
ReactivityMouse, Rat