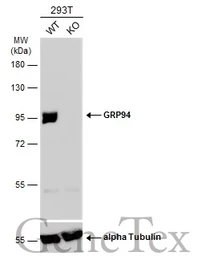
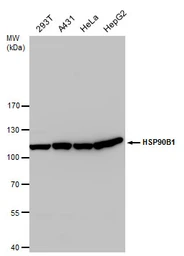
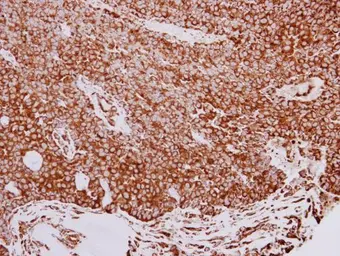
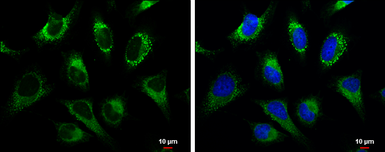
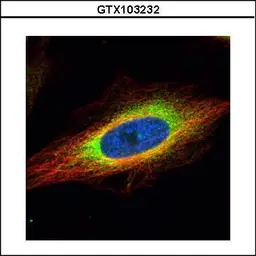
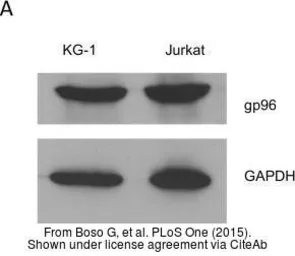
GRP94 antibody
Cat. No. GTX103232
Cat. No. GTX103232
-
HostRabbit
-
ClonalityPolyclonal
-
IsotypeIgG
-
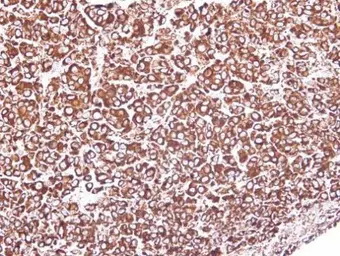
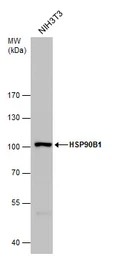
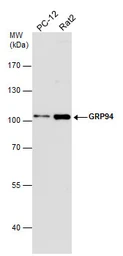
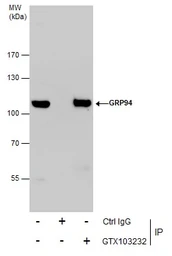
ApplicationsWB ICC/IF IHC-P IP
-
ReactivityHuman, Mouse, Rat