Rat TGF beta 2 protein, His tag
Cat. No. GTX00377-pro
Cat. No. GTX00377-pro
-
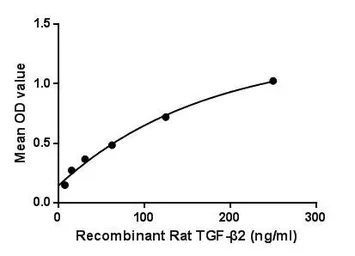
ApplicationsFunctional Assay
-
SpeciesRat